Hansen Solubility Parameters (HSP)
Smart and predictive ingredient matching
- Fast, practical and accurate HSP determinations
- Sustainable and predictive formulation data for all types of ingredients
- Implementation of HSP in formulations
- Match or replace ingredients
- Improve solubility, compatibility, stability and efficacy
- Applicable in solutions, dispersions and blends
How it works
Together with Prof. Steven Abbott and Dr. Charles Hansen, VLCI promotes HSP for a wide range of applications.
HSP Principle
Solubility of a product is represented as a sphere and characterized by three Hansen Solubility Parameters (HSP): 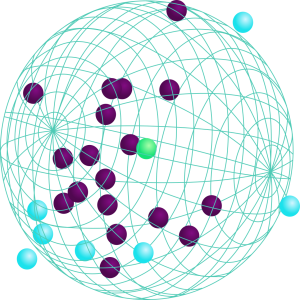
- δD for Dispersion (van der Waals)
- δP for Polarity (Dipole Moment)
- δH for Hydrogen Bonding
The HSP theory is based on the “like seeks like” principle, with likeness measured by the HSP distance metric. The critical conditions for solubility are found within a spherical region of HSP space, around the centre. Therefore, inclusions in the sphere, or overlapping spheres, predict solubility or compatibility.
HSP determination is a powerful and practical way to understand, predict and improve:
- Solubility
- Compatibility
- Stability
- Efficacy
HSPs allow strong predictions for smart and efficient ingredient matching, and is applicable to many types of ingredients: polymers, oils, pigments, solvents, additives…
Our Expertise
VLCI is a certified research centre for determinations of HSP and implementations thereof. The solubility (or dispersibility) of a product is assessed within a standardized set of solvents with known HSPs, spread over the 3-dimensional HSP space. Solvents and products are automatically dispensed via the gravimetric dispensing unit of our High Throughput platform. The solubility is ranked from 1 (excellent) to 6 (very poor). Based on these scores, the HSPiP software generates the solubility sphere of the tested product.
Digitalization of formulation R&D via HSP webapp PrediMatch
Since HSP is a fixed predictive parameter of ingredients to predict other matching, replacing and bridging ingredient to formulate solutions, dispersion and to complement emulsions, it can be sustainbly used, enabling digitalization. See more on this topic via PrediApps section and via PrediApps website to request a demo.
Read Also
Bio-based HSP Determination for Cosmetic Applications
Bio-based HSP Determination for Adhesive Applications
Using HSP to Select Coalescents and Improve Film Formation
Flavor Scalping of a Bio-based Polymer Predicted by HSP
How to Predict Efficient Active Delivery in Cosmetics Using HSP
Determining Solubility of Nail Varnishes via HSP
Practical Determination and Application of HSP for the Paints and Coatings Industry
Using HSP to Improve the Dispersibility of Pigments and Fillers
Watch our tutorials on SpecialChem
Formulate Faster and Smarter with Hansen Parameters – Practical Guide
How to Efficiently Replace Solvents for Nail Polish Removal with HSP
How to Select Pigment/Filler Dispersants Faster using HSP
How to Predict Efficient Active Delivery for Enhanced Efficacy
Let us know how we can help
"*" indicates required fields
Applications & Project Examples
- Solvents outside the solubility sphere of a product can be blended to reach HSP’s within the sphere. By this means, toxic or environmentally unsustainable solvents can be replaced by blends of non-hazardous and/or bio-based solvents.
- Dispersants can be matched with specific solid particles for the preparation of optimally stabilized suspensions.
- Film formation of acrylic dispersions can be enhanced via the effective selection of coalescing agents.
- Solubility of pharmaceutical actives can be significantly improved.
VLCI offers HSP expertise combined with High Throughput capabilities to deliver fast, practical and accurate results.